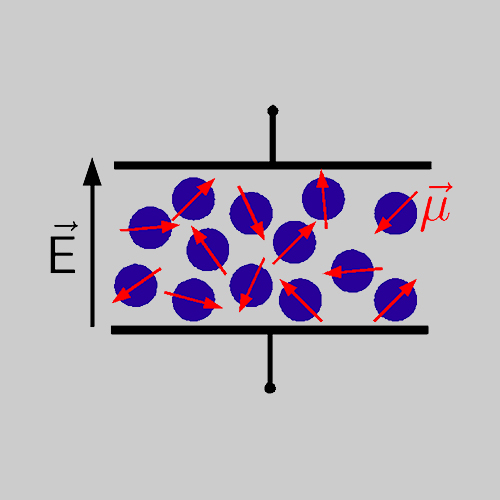
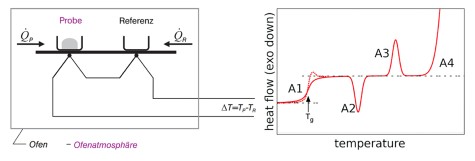
Since the macroscopic dielectric permittivity results from the superposition of all microscopic dipole moments of the molecules in a sample, information regarding molecular dynamics in e.g. simple molecular liquids or mixtures can be gained by analyzing BDS data.
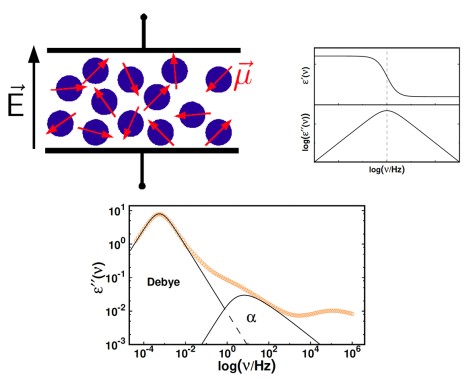
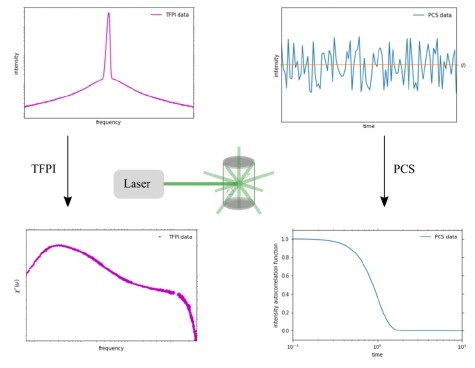
Typically, in experiments an oscillating electric field with frequency ν is applied to the sample and the frequency is varied in the range 10−2 Hz ≤ ν ≤ 107 Hz. This procedure yields the frequency dependent and complex dielectric permittivity ˆε(ν) = ε′(ν) − i ε′′(ν). Here, the real part describes the reversible portion of the permittivity and the imaginary part reflects the loss part. In case the oscillatory frequency of the electric field coincides with the resonance frequency of the molecules inthe sample, one observes a decay in ε′(ν) and a peak in ε′′(ν), as shown in the figure.
In supercooled liquids, BDS allows to identify the different relaxation processes and their respective temperature dependence. E.g. for monohydroxy alcohols, the spectra contain the α-process at moderate frequencies, reflecting the reorientation of single molecules, and a slow Debye process at low frequencies, that can be attributed to the reorientation of supramolecular structures, built via hydrogen bonds.
Additionally, fast relaxation contributions with small amplitudes are observed at high frequencies.In addition to experiments in the frequency range 10−2 Hz ≤ ν ≤ 107 Hz, time-domain experiments((10−6 Hz ≤ ν ≤ 101 Hz)) and high-frequency techniques (108 Hz ≤ ν ≤ 1012 Hz) can be applied. This broadband-character of BDS is particularly valuable, as it allows the characterization of relaxation processes in liquids from temperatures close to the boiling point down to deep into the glass.